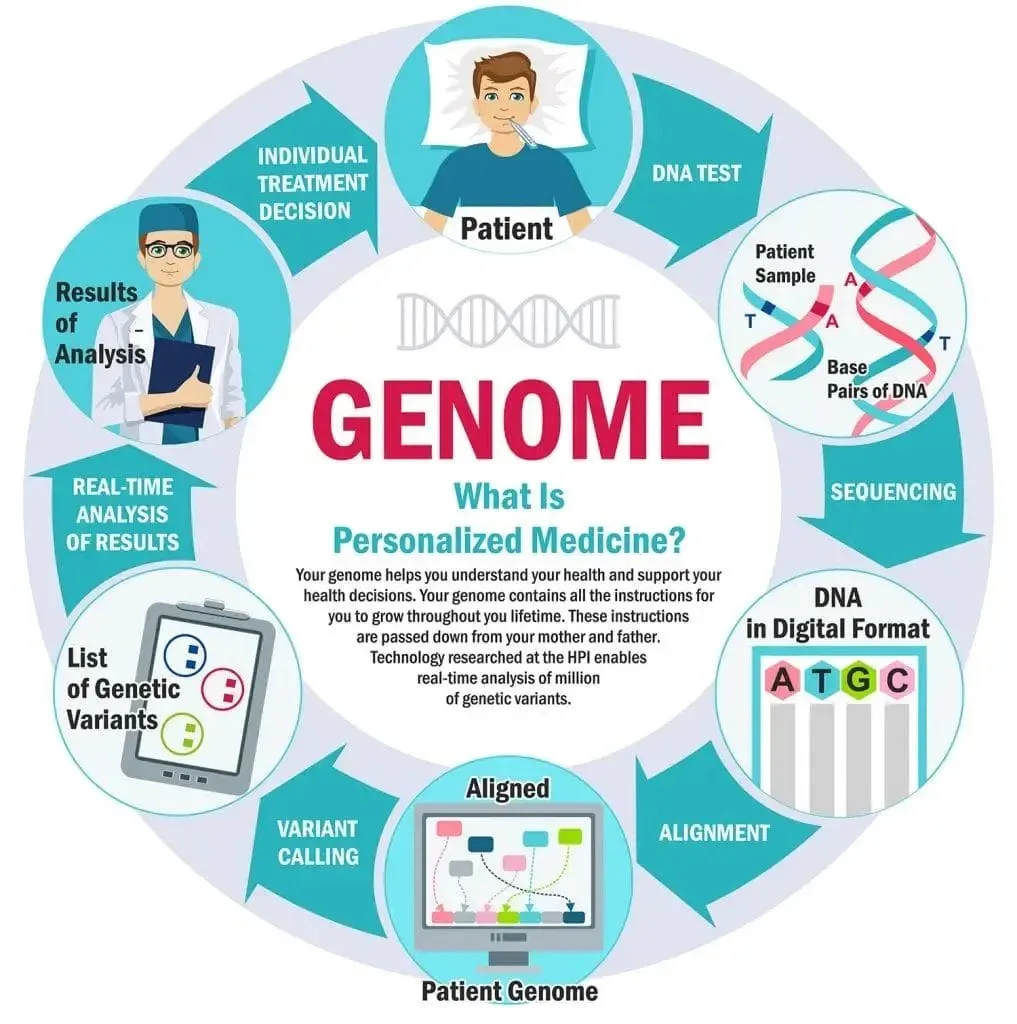
Personalised medicine and genomics are reshaping healthcare by turning data, biology, and digital tools into real-time clinical decisions. This movement links core concepts with advances in genome science to tailor prevention and treatment to the unique biology of each patient. Advances in genomic sequencing and diagnostics are expanding our ability to predict risk, diagnose earlier, and guide therapies. Clinicians, researchers, and health systems are adopting these tools to move away from a one-size-fits-all approach toward care that respects individual genetic profiles. As access to data grows and interpretation improves, the potential to improve outcomes while reducing unnecessary interventions becomes increasingly clear.
Viewed through the lens of tailored medicine and genome-guided care, this shift emphasizes treatments customized to a person’s unique molecular profile. Clinicians increasingly rely on DNA and RNA information, multi-omics data, and AI-driven analysis to inform diagnostics and therapy choices. In the wider lexicon, the movement is often described as precision health or customized care, underscoring predictive, preventive, and personalized decision-making. As adoption grows across clinics, researchers, and policymakers, interoperable data systems and robust evidence generation become essential to prove value and drive sustainable implementation.
Personalised medicine and genomics: from one-size-fits-all to individualized care
Health care is entering a new era where Personalised medicine and genomics push care beyond one-size-fits-all. By integrating genetic, molecular, environmental, and lifestyle data, clinicians can tailor prevention and treatment to the patient’s unique biology. This shift aligns with the broader movement toward precision medicine, leveraging genomics breakthroughs to understand how individuals respond to therapies.
As laboratories generate richer data, doctors can move from generic guidelines to personalized plans that reflect a patient’s genomic risk and biomarkers. Genomic sequencing and other genomic analyses feed into real-world evidence that informs decisions at the point of care, supporting diagnostics, risk assessment, and monitoring.
The role of genomic sequencing in modern diagnostics and therapy
Genomic sequencing has dramatically reduced the cost and time to read a person’s genome, and it is now a core part of cancer diagnostics, rare disease workups, and even some newborn screening programs.
This capability accelerates precision medicine by identifying actionable targets and informing the choice of targeted therapies, which can improve outcomes and help predict prognosis.
Multi-omics and precision medicine: building the full biological picture
Multi-omics profiling integrates genomic, transcriptomic, proteomic, and metabolomic data to create a fuller model of disease biology for precision medicine.
This holistic view supports more accurate patient stratification and helps clinicians select therapies with higher likelihoods of success while minimizing adverse effects.
Pharmacogenomics and tailored drug therapy
Pharmacogenomics studies how genes influence drug metabolism and response, guiding dosing and drug choice in a way that aligns with personalised medicine. Genetic testing in healthcare can reveal variants that affect safety and effectiveness, enabling tailored prescriptions.
In oncology and cardiology, pharmacogenomic insights help select targeted drugs and adjust doses to reduce adverse events.
AI, real-world evidence, and decision support for personalised care
AI and real-world evidence turn complex genomic data into practical clinical insights, enabling faster interpretation and safer decisions.
Decision-support systems flag risks, suggest targeted therapies, and help integrate results into routine workflows, driven by robust data analytics and interoperable electronic health records.
Ethics, equity, policy, and the path forward for sustainable adoption
As genomic tools scale, privacy, consent, and data governance become central concerns for genetic testing in healthcare.
Policy actions—education for clinicians, transparent pricing, reimbursement models, and public-private partnerships—will be essential to ensure equitable access and to sustain innovation in personalised medicine and genomics.
Frequently Asked Questions
How do personalised medicine and genomics work together to tailor prevention, diagnosis, and treatment?
Personalised medicine uses an individual’s genetic, molecular, environmental, and lifestyle data to tailor care, while genomics provides the blueprint by revealing genetic variants that influence disease risk and therapy response. Together, they enable precision diagnostics and targeted prevention and treatment strategies, moving beyond a one size fits all approach.
How does genomic sequencing enable precision medicine in cancer care?
Genomic sequencing reads tumor and germline DNA to identify driver mutations and actionable targets, guiding targeted therapies, improving diagnostic accuracy, and enabling personalized treatment plans within precision medicine.
What is pharmacogenomics and how does it influence drug prescriptions in personalised medicine?
Pharmacogenomics studies how genetic differences affect drug metabolism and response. By incorporating genetic variants into prescribing decisions, clinicians can adjust doses or choose alternatives, a key practice in personalised medicine and precision medicine.
How can genetic testing in healthcare aid in diagnosing rare diseases and guiding management?
Genetic testing in healthcare, including genomic sequencing, helps uncover pathogenic variants responsible for rare conditions, leading to earlier diagnosis, tailored management, and informed family planning and genetic counseling.
What ethical and policy considerations arise with genomics breakthroughs in healthcare?
Genomics breakthroughs raise concerns about data privacy, informed consent, equitable access, and representation in research. Addressing these issues is essential to ensure responsible use of personalised medicine and genomics.
What barriers exist to implementing genomics breakthroughs in routine care, and how can they be overcome?
Barriers include data fragmentation, limited clinician genomics training, and the need for scalable bioinformatics and interoperable health records. Solutions include education, cloud-based analytics, secure data sharing, and public-private partnerships to accelerate adoption in personalised medicine.
| Aspect | Key Point | Examples/Notes |
|---|---|---|
| Definition | Personalised medicine uses individual biological, environmental, and lifestyle data to guide care; Genomics provides the blueprint for why people respond differently to treatments. | A shift from generic care to care tailored to each patient. |
| Spectrum shift | Medicine is a spectrum from broad public guidelines to precise, individual interventions; driven by sequencing, data analytics, and real‑world evidence. | Leads to new, targeted therapies, diagnostics, and monitoring tools. |
| Breakthrough: High-throughput sequencing | Reads a person’s genome cost- and time-efficiently. | Used in cancer diagnostics, rare disease workups, and some newborn screening; enables actionable targets. |
| Breakthrough: Multi-omics profiling | Integrates genomic, transcriptomic, proteomic, and metabolomic data. | Improves patient stratification and helps select therapies with higher success and fewer side effects. |
| Breakthrough: Pharmacogenomics | Genes influence how drugs are metabolized and respond. | Guides dosing or drug choice to improve safety and effectiveness; used in oncology and beyond. |
| Breakthrough: AI and computational tools | Advanced analytics convert complex genomic data into usable clinical insights. | Decision-support, risk flags, and therapy suggestions integrated into workflows. |
| Clinical applications | Broad use from diagnosis to treatment | Oncology tumor profiling; metabolic/inherited diseases; cardiology/neurology pharmacogenomics; immunology/infectious disease; differential diagnosis; genetic counseling for families. |
| Ethics, equity, and policy | Data privacy, governance, consent, and ensuring equitable access; interoperability and evidence generation. | Protect privacy while enabling research and clinical use; include diverse populations; standardize data and generate real-world evidence. |
| Barriers and Path Forward | Data fragmentation, limited clinician genomics training, and scalable bioinformatics infrastructure. | Expand genomics education; invest in cloud-based analytics; public-private partnerships; promote patient engagement and transparent deployment. |
Summary
Conclusion: Personalised medicine and genomics is transforming health by linking genomic insights with clinical care to tailor prevention, diagnosis, and treatment. This shift supports more predictive, preventive, and precise care, with potential for better outcomes and patient satisfaction, while highlighting the need to address privacy, ethics, and equity. Realizing this potential will require ongoing collaboration among patients, clinicians, researchers, policymakers, and industry to build robust data governance, scalable infrastructure, and equitable access to genomic tools.